BSEB Bihar Board Class 10 Science Solutions Chapter 2 Acids, Bases and Salts Textbook Questions and Answers.
BSEB Bihar Board Class 10 Science Solutions Chapter 2 Acids, Bases and Salts
Bihar Board Class 10 Science Chapter 2 Acids, Bases and Salts InText Questions and Answers
In-text Questions (Page 18)
Question 1.
You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic solution and a basic solution respectively. If you are given only red litmus paper, how will you identify the contents of each test tube?
Answer:
Dip red litmus paper strips separately in all the three test tubes. The test tube in which red litmus strip turns blue, contains basic solution. Now take the blue litmus paper and dip in the remaining test tubes. If the colour of the blue litmus paper changes to red, the test tube contains acidic solution. In case it remains blue then the test tube contains distilled water.
In-text Questions (Page 22)
Question 1.
Why should curd and sour substances not be kept in brass and copper vessels?
Answer:
Curd and sour substances are not stored in brass and copper vessels because they are acidic in nature. The acid present in them react with metals to form toxic compounds which are poisonous for humans.
Question 2.
Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test the presence of this gas?
Answer:
When a metal reacts with an acid, usually hydrogen gas is liberated. For example, zinc reacts with dilute sulphuric acid as follows:
![]()
The hydrogen gas evolved can be tested by passing the gas through soap solution and then bringing a burning candle near the rising soap bubble filled with the gas. The soap bubble bursts and the hydrogen gas burns with a pop sound.
Question 3.
Metal compound A reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride.
Answer:
The compound ‘A’ is calcium carbonate. The gas evolved is carbon dioxide which extinguishes a burning candle.

In-text Questions (Page 25)
Question 1.
Why do HCl, HNO3, etc. show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?
Answer:
Although alcohol and glucose contain hydrogen, they do not ionise in solution to produce H+ ions, which are responsible for the acidic properties of a substance. On the other hand, HCl and HNO3 furnish H+ ions in solution and thus, show acidic property.
Question 2.
Why does an aqueous solution of an acid conduct electricity?
Answer:
Aqueous solution of an acid releases H+ ions or H3O+ ions and anions (A–) in solution. Since ions are the carrier of charge, the aqueous solution of an acid conducts electricity.
Question 3.
Why does dry HCl gas not change the colour of the dry litmus paper?
Answer:
Dry HCl gas does not ionise to produce H+ ions. Hence, it does not show acidic properties and, therefore, does not change the colour of dry litmus paper.
Question 4.
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid? [CBSE 2011]
Answer:
Acids, particularly the mineral acids like H2SO4, HNO3 and HCl, etc., have strong affinity for water. The dilution process is highly exothermic in nature. The heat evolved may convert the water to steam or may crack or break the container and the acid water mixture may splash out. If these come in contact with the skin of the person handling the acid, he may suffer from severe burns. Therefore it is advised to add acid drop by drop to water. So that the heat is dissipated evenly in water.
Question 5.
How is the concentration of hydronium ions (H3O+) affected when a solution of an acid is diluted?
Answer:
The concentration of hydronium ions (H3O+) decreases per unit volume on diluting an acid, i.e., the solution becomes less acidic on dilution.
Question 6.
How is the concentration of hydroxide (OH–) ions affected when excess of base is dissolved in a solution of sodium hydroxide?
Answer:
Sodium hydroxide (NaOH) is a strong base. It immediately dissociates in solution to give OH– ions and Na+ ions. Upon dissolving more of base in this solution, the concentration of OH– ions further increases.
In-text Questions (Page 28)
Question 1.
You have two solutions A and B. The pH of the solution A is 6 and pH of solution B is 8. Which solution has more hydrogen ion concentration? Which of this is acidic and which one is basic?
Answer:
A pH value of less than 7 indicates an acidic solution, while greater than 7 indicates a basic solution. Therefore, the solution with pH = 6 is acidic and has more hydrogen ion concentration than the solution of pH = 8 which is basic.
Question 2.
What effect does the concentration of H+(aq) ions have on the nature of the solution?
Answer:
The concentration of hydrogen ions is directly related to the acidic nature of the solution. Higher is the concentration of H+ ions in the solution, more acidic is the solution.
Question 3.
Do basic solutions also have H+(aq) ions? If yes, then why are these basic?
Answer:
Yes, the basic solutions also have H+ ions. They are basic because OH– ions are more than H+ ions.
Question 4.
Under what soil condition do you think a farmer would spread or treat the soil of his fields with quick lime (calcium oxide) or slaked lime (calcium hydroxide) or chalk (calcium carbonate)?
Answer:
For healthy growth of plants, the soil should neither be alkaline nor acidic. As quick lime or slaked lime or chalk are all alkaline substances, these would be added to an acidic soil by the farmer.
In-text Questions (Page 33)
Question 1.
What is the common name of the compound CaOCl2?
Answer:
Bleaching powder.
Question 2.
Name the substance which on treatment with chlorine yields bleaching powder.
Answer:
Slaked lime Ca(OH)2 [Calcium hydroxide].
Question 3.
Name the sodium compound used for softening hard water.
Answer:
Washing soda or sodium carbonate (Na2CO3. 10H2O).
Question 4.
What will happen if the solution of sodium hydrogen carbonate is heated? Give the equation of the reaction involved.
Answer:
Sodium hydrogen carbonate on heating decomposes to gives sodium carbonate, carbon dioxide and water.

Question 5.
Write an equation to show the reaction between Plaster of Paris and water.
Answer:

Bihar Board Class 10 Science Chapter 2 Acids, Bases and Salts Textbook Questions and Answers
Question 1.
A solution turns red litmus blue, its pH is likely to be
(a) 1
(b) 4
(c) 5
(d) 10
Answer:
(d) Solution is basic, so pH is likely to be 10.
Question 2.
A solution reacts with crushed egg-shells to give a gas that turns lime water milky. The solution contains
(a) NaCl
(b) HCl
(c) LiCl
(d) KCl
Answer:
(b) HCl
Question 3.
10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount HCl solution (the same solution as before) required to neutralise it will be
(a) 4 mL
(b) 8 mL
(c) 12 mL
(d) 16 mL
Answer:
(d) As per neutralisation double amount of HC1 will be required i.e., 8 × 2 = 16 mL.
Question 4.
Which one of the following types of medicines is used for treating indigestion?
(a) Antibiotic
(b) Analgesic
(c) Antacid
(d) Antiseptic
Answer:
(c) Antacid is used in indigestion.
Question 5.
Write word equations and then balanced equations for the reactions taking place when:
(a) dilute sulphuric acid reacts with zinc granules.
(b) dilute hydrochloric acid reacts with magnesium ribbon.
(c) dilute sulphuric acid reacts with aluminium powder.
(d) dilute hydrochloric acid reacts with iron fillings.
Answer:
(a) Word equation: Zinc + Sulphuric acid → Zinc sulphate + Hydrogen
Balanced equation: Zn(s) + H2SO4(dil.) → ZnSO4(aq) + H2(g)
(b) Word equation: Magnesium + Hydrochloric acid → Magnesium chloride + Hydrogen
Balanced equation: Mg(s) + 2HCl(dil.) → MgCl2(aq) + H2(g)
(c) Word equation: Aluminium + Sulphuric acid → Aluminium sulphate + Hydrogen
Balanced equation: 2Al(s) + 3H2SO4(dil.) → Al2(SO4)3(aq) + 3H2(g)
(d) Word equation: Iron + Hydrochloric acid → Iron chloride + Hydrogen
Balanced equation: Fe(s) + 2HCl(dil.) → FeCl2(ag) + H2(g)
Question 6.
Compounds like alcohol and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
Answer:
The chemical formula of ethyl alcohol is C2H5OH and that of glucose is C6H12O6. Both are organic compounds containing hydrogen atoms, but these do not dissociate in water to give H+ or H3O+ ions.
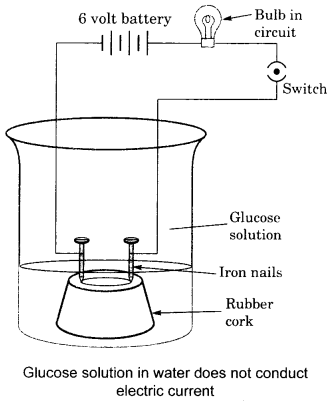
This can be shown by the following activity:
In a glass beaker, take a dilute solution of glucose (C2H12O6). Fix two small iron nails in a rubber cork and place the cork in a beaker as shown in the figure. Connect the nails to the terminals of a 6 volt battery through a bulb. Switch on the current. The bulb will not glow. This shows that the electric current has not passed through the glucose solution. As the current is carried by the movement of ions, this shows that glucose solution, does not dissociate to produce H+ ions.
Now repeat the same experiment with ethyl alcohol (C2H5OH). The bulb will not glow in this case also. This shows that both of these compounds do not behave as acids although they contain hydrogen atoms in their molecules.
Question 7.
Why does distilled water not conduct electricity, whereas rain water does?
Answer:
Distilled water is pure water, does not dissociate into ions because it is a weak electrolyte. Salts are present in rainwater which help in dissociation of rainwater into ions. These ions helps in conduction of electricity.
Question 8.
Why do acids not show acidic behaviour in the absence of water?
Answer:
Because acids like HCl does not dissociate into ions in the absence of water.
Question 9.
Five solutions A, B, C, D and E when tested with an universal indicator showed pH as 4, 1, 11, 7 and 9 respectively. Which solution is
(а) neutral
(b) strongly alkaline
(c) strongly acidic
(d) weakly acidic
(e) weakly alkaline?
Arrange the pH in increasing order of hydrogen-ion concentration.
Answer:
(a) Solution D with pH 7 is neutral.
(b) Solution C with pH 11 is strongly alkaline.
(c) Solution B with pH 1 is strongly acidic.
(d) Solution A with pH 4 is weakly acidic.
(e) Solution E with pH 9 is weakly alkaline.
Increasing order of hydrogen ion concentration: C < E < D < A < B
Question 10.
Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. Amount and concentration taken for both the acids are same. In which test tube will the fizzing occur more vigorously and why?
Answer:
More vigorous fizzing will occur in test tube A containing hydrochloric acid (HCl). This is because hydrochloric acid is stronger than acetic acid. The reaction of magnesium with hydrochloric acid to give out hydrogen gas is faster than with acetic acid.

Question 11.
Fresh milk has a pH of 6. How do you think the pH will change as it turns into curd? Explain your answer.
Answer:
On conversion of milk into curd, its pH will decrease and will become less than 6. The pH decreases as curd is more acidic than milk due to the presence of lactic acid. More acidic is the substance, lesser is its pH.
Question 12.
A milkman adds a very small amount of baking soda to fresh milk.
(а) Why does he shift the pH of the fresh milk from 6 to slightly alkaline?
(b) Why does this milk take a long time to set as curd? [CBSE 2011]
Answer:
(a) We know that fresh milk is slightly acidic due to the presence of lactic acid and its pH is 6. Upon standing, its pH slowly decreases and it becomes sour since more of H+ ions is released. The purpose of adding baking soda or sodium hydrogencarbonate (NaHCO3) is to make the medium slightly alkaline. The OH– ions released neutralise the effect of lactic acid present in milk. This checks the milk from getting sour.
(b) When milk sets as curd, it becomes more acidic and its pH decreases. In the alkaline medium, it takes longer time to achieve acidic medium back so that it may set as curd.
Question 13.
Plaster of Paris should be stored in a moisture proof container. Explain why?
Answer:
Plaster of Paris (CaSO4. \(\frac {1}{2}\)H2O) has a strong tendency to absorb moisture and get converted into gypsum (CaSO4. 2H2O) which sets into a hard mass. Thus, to prevent its contact with moisture, it is stored in a moisture proof container.
Question 14.
What is a neutralisation reaction? Give two examples.
Answer:
A chemical reaction in which an acid reacts with a base to form salt and water, is called neutralisation reaction. For example,

Question 15.
Give two important uses of washing soda and baking soda.
Answer:
Uses of washing soda
- In the manufacture of glass, soap, paper and chemicals like caustic soda (NaOH) and borax (Na2B4O7), etc.
- As a cleansing agent for domestic purposes.
Uses of baking soda:
- In baking powder used for preparing cakes.
- In antacids to reduce acidity in the stomach.
Bihar Board Class 10 Science Chapter 2 Acids, Bases and Salts Textbook Activities
Activity 2.1 (Textbook Page 18)
Observation Table:

Conclusion:
- Acids turn blue litmus red but have no effect on red litmus.
- Bases turn red litmus blue but have no effect on blue litmus.
- Phenolphthalein is colourless in acidic medium and turns pink in basic medium,
- Methyl orange is yellow in basic medium and red in acidic medium.
Activity 2.2 (Textbook Page 18)
Observation:

Conclusion: Olfactory indicators like onion juice, vanilla essence, clove oil lose their smell in basic medium.
Activity 2.3 (Textbook Page 19)
Observation:
- When zinc granules are dropped in the test tube containing dilute sulphuric acid, effervescence occurs and bubbles stick to the surface of the granules indicating the formation of a gas.
- On passing the gas through the soap solution, soap bubbles filled with gas, fly out and burst in air.
Conclusion:
- Bubbles are formed in soap solution indicating the formation of a gas.
- The gas burns with a pop sound.
- The gas evolved is hydrogen.
- The same observation is found in all the cases, only the evolution of hydrogen gas is slow with weak acids
like CH3COOH.
Conclusion: Zinc displaces hydrogen from all the acids and forms the corresponding salt.
Metal + Acid → Salt + Hydrogen gas
Activity 2.4 (Textbook Page 20)
Observation: The reaction proceeds with effervescence and bubbles of gas can be seen to escape from the solution. The gas burns with a pop sound. Zinc react with strong bases like NaOH, KOH etc. to liberate hydrogen gas and form the corresponding salt. For example,
![]()
Activity 2.5 (Textbook Page 20)
Observation: Carbon dioxide gas is evolved in each case which turns lime water milky.

On passing excess CO2, the solution becomes clear due to formation of soluble calcium

Conclusion: All metal carbonates and hydrogen carbonate react with acids to form the corresponding salt, carbon dioxide and water.
Metal carbonate/ Metal hydrogen carbonate + Acid → Salt + Carbon dioxide + Water
Activity 2.6 (Textbook Page 21)
Observation:
- On adding phenolphthalein to NaOH solution, it becomes pink.
- On adding dilute HCl solution dropwise, the pink colour disappears and the solution again becomes colourless. This is because the medium becomes acidic.
- On adding NaOH, pink colour reappears because the medium becomes basic again.
Conclusion: Phenolphthalein turns pink in a basic solution and colourless in an acidic solution.
Acid + Base → Salt + Water
Activity 2.7 (Textbook Page 21)
Observation: Black copper oxide dissolves and a blue-green solution is obtained. The blue-green solution is obtained due to the formation of copper (II) chloride in solution.
Conclusion: Copper oxide reacts with hydrochloric acids to form copper (II) chloride, a salt and water. e.g.,

Activity 2.8 (Textbook Page 22)
Observation: The bulb glows in case of acids but not in case of glucose and alcohol solutions.
Conclusion: Electric current is carried through the solution by ions. This shows that acids dissociate in solutions to produce H+ ions but substances like glucose and alcohol do not dissociate to produce any ion.
Activity 2.9 (Textbook Page 23)
Observation: Dry blue litmus paper does not change colour but wet blue litmus paper changes its colour to red. Conclusion: Dry HCl gas is not acidic but HCl solution is acidic. This is because in the presence of water, HCl dissociates to give H+ ions.
![]()
Activity 2.10 (Textbook Page 24)
Observation: In both cases, the temperature is found to rise.
Conclusion: Dilution of concentrated sulphuric acid (H2SO4) with water as well as dissolution of sodium hydroxide (NaOH) in water is exothermic.
Activity 2.11 (Textbook Page 26)
Observation: Saliva (before meal)-[pH = 7.4] [colour of pH paper = green]; Saliva (after meal)-[5 to 6] [yellow to green]; Lemon juice-(2.2) (orange); Colourless aerated drinks-(6.0) (light green); Carrot juice-(6.0) (light green); Coffee (5.0) (greenish yellow); Tomato juice (4.1) light orange); Tap water (7.0) (green); 1 M NaOH (14) (dark purple); 1 M HCl (1.0) (red).
Conclusion: Solution with pH < 7 but close to 7 are weakly acidic. Solution with pH << 7 are strongly acidic. Solutions with pH > 7 but close to 7 are weakly basic. Solutions with pH >> 7 are strongly basic.
Activity 2.12 (Textbook Page 27)
Observation: The ideal soil pH should be close to 7, i.e., neither very acidic nor very alkaline.
Activity 2.13 (Textbook Page 28)
Observation and conclusion:

Families:
(i) On the basis of common acids: Sulphates = K2SO4, Na2SO4, CaSO4, MgSO4, CuSO4; Carbonates = Na2CO3; Chlorides = NaCl, NH4Cl;
(ii) On the basis of common bases: Sodium salts = Na2SO4, NaCl, NaNO3, Na2CO3, Potassium salts = K2SO4 Calcium salts = CaSO4, Magnesium salts = MgSO4, Copper salts = CuSO4, Ammonium salts = NH4Cl.
Activity 2.14 (Textbook Page 29)
Observation:

All the salts are soluble in water.
Conclusion: Salts of a strong acid and a strong base are neutral with pH = 7.
Salts of a strong acid and weak base are acidic with pH < 7 and those of a strong base and weak acid are basic with pH > 7.
Activity 2.15 (Textbook Page 32)
- Blue coloured copper sulphate crystal on heating leave behind white anhydrous copper sulphate and water droplets are seen in the upper cooler parts of the boiling tube.
- On adding 2-3 drops of water to the white residue, blue colour reappears.
Conclusion: Copper sulphate crystals on heating lose water to form white anhydrous copper sulphate which combines with water to from blue coloured copper sulphate crystals.