BSEB Bihar Board Class 10 Science Solutions Chapter 4 Carbon and Its Compounds Textbook Questions and Answers.
Bihar Board Class 10 Science Solutions Chapter 4 Carbon and Its Compounds
Bihar Board Class 10 Science Chapter 4 Carbon and Its Compounds InText Questions and Answers
In-text Questions (Page 61)
Question 1.
What would be the electron dot structure of carbon dioxide which has the formula CO2? [CBSE 2013]
Answer:
Atomic number of carbon = 6
Electronic configuration of carbon = \(\begin{array}{rl}
\mathrm{K} & \mathrm{L} \\
2 & 4
\end{array}\)
Atomic number of O = 8
Electronic configuration of oxygen = \(\begin{array}{rl}
\mathrm{K} & \mathrm{L} \\
2 & 6
\end{array}\)
So carbon needs four electrons more and oxygen needs two more for completion of octet. Hence, carbon shares two of its electrons with two electrons of one oxygen atom and remaining two with two electrons of other oxygen atom.
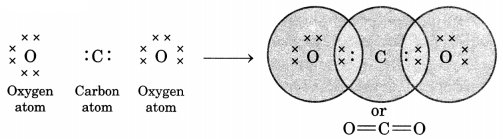
Thus, each oxygen is bonded to the carbon atom by a double bond.
Question 2.
What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur?
Answer:
The atomic number (Z) of sulphur is sixteen and its electronic configuration is 2, 8, 6. Sulphur atom has sixteen electrons. The chemical formula of sulphur molecule is S8. Each sulphur atom is linked to similar atoms on either sides by single covalent bonds and thus, completes its octet. The molecule is in the form of a ring also represented by a crown shape.

In-text Questions (Page 68-69)
Question 1.
How many structural isomers can you draw for pentane?
Answer:
Pentane (C5H12) has a skeleton of five carbon atoms. These five carbon atoms can exist as a straight chain as well as in branched chains. There are three structural isomers for the hydrocarbon which is an alkane.

Question 2.
What are the two properties of carbon which lead to the huge number of carbon compounds we see around us? [CBSE 2013, 2014]
Answer:
The two main properties which leads to huge number of carbon compounds are:
(i) Catenation: It is self linking property of carbon, due to it’s small size and high electronegativity. As a result, each carbon becomes capable of forming stable bonds with other carbon atoms. This can be extended in the form of straight chain or branched chains or in the form of rings.
(ii) Tetravalency of carbon: Carbon having four electrons in its valence shell, can share these four electrons either with the other four carbon atoms or with any other elements like N, O, H, S and halogens, etc. Hence, the number of carbon compounds increases to a large extent.
Question 3.
What will be the formula and electron dot structure of cyclopentane?
Answer:

The general formula of cycloalkanes is CnH2n. Putting n = 5 in this general formula, the formula of cyclopentane is C5H2 × 5 = C5H10.
Now carbon has 4 electrons in the valence shell and hydrogen has one. Therefore, to complete its octet, each carbon shares two of its electrons, one each with two other carbons atoms forming a five-member ring. The remaining two electrons of each carbon share one electron each with two hydrogen atoms. In this way, each hydrogen completes its duplet. The complete electron dot structure of cyclopentane is shown alongside.
Question 4.
Draw the structures for the following compounds:
Are structural isomers possible for bromopentane?
(i) Ethanoic acid
(ii) Bromopentane
(iii) Butanone
(iv) Hexanal
Answer:


Bromopentane has a chain of five carbon atoms. It can exist in a number of forms which are structural isomers.


- The structural isomers (i), (ii) and (iii) which differ in the position of the Br atom are known as position isomers.
The structural isomers (iv), (v) and (vi) which differ in the arrangement of carbon atoms in the chain are called chain isomers. - In writing the IUPAC name, the name of prefix bromo is written before that of prefix methyl. In fact, alphabetical order is followed while naming the different prefixes.
Question 5.
How would you name the following compounds? [CBSE 2014]

Answer:
Name of following compounds:

In-text Questions (Page 71)
Question 1.
Why is the conversion of ethanol into ethanoic acid an oxidation reaction?
Answer:
Ethanoic acid (CH3COOH) has one oxygen atom more and two hydrogen atoms less than ethanol (C2H5OH).
In general C2H5OH → CH3COOH
- Loss of hydrogen is known as oxidation.
- Gain of oxygen is known as oxidation.
Therefore, this conversion is an oxidation reaction.
Question 2.
A mixture of oxygen and ethyne is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Answer:
When mixture of oxygen and ethyne is burnt, it produces a high temperature because of complete combustion of the hydrocarbon and gives a clean flame. But when ethyne is burnt in air, it results in incomplete combustion. Incomplete combustion will not give a high temperature simultaneously it will produce a sooty flame.
In-text Questions (Page 74)
Question 1.
How would you distinguish experimentally between an alcohol and a carboxylic acid? [CBSE 2013]
Answer:

Question 2.
What are oxidising agents?
Answer:
Oxidising agents are the substances which either on their own or on reacting with another chemical substances release oxygen in order to carry out oxidation reactions. The commonly used oxidising agents are: Ozone, bromine water, a mixture of potassium permanganate [KMNO4] and sulphuric acid, etc.
In-text Questions (Page 76)
Question 1.
Would you be able to check if water is hard by using a detergent?
Answer:
Detergent gives lather with hard and soft water both, while a soap gives lather with soft water only. Thus, it is not possible to check if water is hard; by using a detergent.
Question 2.
People use a variety of methods to wash clothes. Usually after adding the soap, they ‘beat’ the clothes on a stone, or beat it with a paddle, scrub with a brush or the mixture is agitated in a washing machine. Why is agitation necessary to get clean clothes?
Answer:
During the cleansing action of soaps and detergents, the hydrocarbon tails of the soaps form an interface between the grease or dirt on the surface of cloth and water. Hence, surface tension of water decreases and a stable mixture of oil in water is formed. To wash away the loosened dirt particles from the surface of the cloth, it is agitated either mechanically, or beaten with a stone or with paddle or agitated in a washing machine.
Bihar Board Class 10 Science Chapter 4 Carbon and Its Compounds Textbook Questions and Answers
Question 1.
Ethane, with the molecular formula C2H6 has [CBSE 2011]
(a) 6 covalent bonds
(b) 7 covalent bonds
(c) 8 covalent bonds
(d) 9 covalent bonds
Answer:

Question 2.
Butanone is a four carbon compound with the functional group
(a) carboxylic acid
(b) aldehyde
(c) ketone
(d) alcohol
Answer:

In butanone, the functional group is ketone.
Question 3.
While cooking, if the bottom of the vessel is getting blackened on the outside, it means that
(a) the food is not cooked completely
(b) the fuel is not burning completely.
(c) the fuel is wet.
(d) the fuel is burning completely.
Answer:
(b) Due to incomplete burning or combustion of fuel.
Question 4.
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Answer:
In CH3Cl i.e., Chloromethane

Since carbon and chlorine needs 4 and 1, electron each respectively to complete their octet and hydrogen needs one electron to complete its duplet, hence carbon shares its four electrons one each, with three hydrogen atoms and one electron with a chlorine atom.

Hence, chloromethane has:
Three C—H covalent bonds
One C—Cl covalent bond
Question 5.
Draw the electron dot structures for:
(a) ethanoic acid
(b) H2S
(c) propanone
(d) F2
Answer:
Electron dot structure for:
(a) Ethanoic acid:

(b) H2S:

(c) Propanone:

(d) F2 (Fluorine molecule):

Question 6.
What is a homologous series? Explain with an example.
Answer:
A homologous series is a series of carbon compounds that have different numbers of carbon atoms but contain the same functional group. For example, methane, ethane, propane, butane, etc., are all parts of the alkane homologous series. The general formula of this series is CnH2n + 2.
Methane CH4
Ethane CH3CH3
Propane CH3CH2CH3
Butane CH3CH2CH2CH3
It can be noticed that there is a difference of CH2 unit between each successive compound.
Question 7.
How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties? [CBSE 2011]
Answer:
Distinction based on physical properties:
- Smell: Ethanol has a characteristic smell known as alcoholic smell which is pleasant. Ethanoic acid has vinegar-like smell.
- Boiling points: Boiling point of ethanol (351 K) is less than that of ethanoic acid (391 K).
- Litmus test: Ethanol is neutral in nature and does not bring any change in the colour of litmus whether blue or red. Ethanoic acid is acidic and changes the colour of a blue litmus strip to red when dipped in it.
Distinction based on chemical properties:
(i) Action with sodium hydrogen carbonate: On adding a small amount of sodium hydrogen carbonate to ethanoic acid, carbon dioxide gas is evolved with brisk effervescence. However, no such reaction . is noticed in case of ethanol.
CH3COOH + NaHCO3 → CH3COONa + CO2 + H2O
(ii) Action with sodium (Na):
On adding a few pieces of sodium to ethyl alcohol, hydrogen gas is evolved. 2Na + 2CH3CH2OH → 2CH3CH2O– Na+ + H2
Question 8.
Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also? [CBSE 2011, 2013]
Answer:
A soap has two parts:
(a) A polar, water-soluble, hydrophilic end.
(b) A non-polar, water-insoluble, hydrophobic end.
So when soap is added to water, polar ends dissolve in water whereas non-polar do not. Hence, spherical structures are formed called micelles.
But in ethanol, soap is soluble, hence micelles are not formed.
Question 9.
Why are carbon and its compounds used as fuels for most applications?
Answer:
Most of the carbon compounds give a lot of heat and light when burnt in air. Saturated hydrocarbons burn with a clean flame and no smoke is produced. The carbon compounds, used as a fuel, have high calorific values. Therefore, carbon and its compounds are used as fuel for most applications.
Question 10.
Explain the formation of scum when hard water is treated with soap.
Answer:
Hard water contains calcium and magnesium ions. These combine with soap molecules to form curdy white precipitate of calcium and magnesium salts of soap which is called scum.

Question 11.
What change will you observe if you test soap with litmus paper (blue and red)?
Answer:
When soap is dissolved in water, the solution is alkaline in nature due to the formation of alkali NaOH or KOH. The solution changes the colour of red litmus to blue. However, the solution does not change the colour of blue litmus.
Question 12.
What is hydrogenation? What is its industrial application?
Answer:
Unsaturated hydrocarbons undergo addition reaction in presence of catalysts such as palladium or nickel to give saturated hydrocarbons. This process is called hydrogenation. Unsaturated vegetable oil is converted to saturated semi-solid material by hydrogenation reaction using nickel catalysts.
Question 13.
Which of the following hydrocarbons undergo addition reactions:
C2H6, C3H8, C3H6, C2H2 and CH4.
Answer:
In order that a hydrocarbon may undergo addition reaction, it must be unsaturated in nature. It must be either an alkene (C=C) with general formula CnH2n or an alkyne (C≡C) with general formula CnH2n-2.
Out of the list of the hydrocarbons given:
• C3H6(Propene) is an alkene with C=C bond. It corresponds to general formula CnH2n (n = 3)
• C2H2 (Ethyne) is an alkyne with C≡C bond. It corresponds to general formula CnH2n-2 (n = 2).
Both these hydrocarbons take part in addition reactions. For example, they react with hydrogen upon heating to 473 K in the presence of Nickel catalyst to form the corresponding alkanes.

Question 14.
Give a test that can be used to differentiate chemically between butter and cooking oil. [CBSE 2013]
Answer:
Butter is a saturated compound, whereas cooking oil is an unsaturated compound. Unsaturated compounds gives positive Baeyer’s Reagent test (test with alkaline KMnO4).

Question 15.
Explain the mechanism of the cleaning action of soaps.
Answer:
Cleansing action of soaps: The dirt present on clothes is organic in nature and insoluble in water. Therefore, it cannot be removed by only washing with water. When soap is dissolved in water, its hydrophobic ends attach themselves to the dirt and remove it from the cloth. Then the molecules of soap arrange themselves in micelle formation and trap the dirt at the centre of the cluster. These micelles remain suspended in the water. Hence, the dust particles are easily rinsed away by water.

Bihar Board Class 10 Science Chapter 4 Carbon and Its Compounds Textbook Activities
Activity 4.1 (Textbook Page 58)
Observation: Except water and cooking utensils (made up of metals, glass/clay or ceramics), a large number of things such as soaps, detergents, newspaper, books. Furniture (made up of wood and plastic) and various food items are made up of compounds of carbon. However, some of the things such as mugs, cups/plates are usually made up of glass or ceramics while buckets are made up of metals or plastics.
Conclusion: Most of the things we use or consume in our day to day life are made up of compounds of carbon.
Activity 4.2 (Textbook Page 67)
Observation:

The difference in formulae in each of these three cases is the same, i.e., CH2 and the difference in molar masses is also the same, i.e., 14 u.
The order of increasing carbon atoms of these four alcohols is:
CH3OH, C2H5OH, C3H7OH, C4H9OH
Yes, these four alcohols constitute a homologous series.
Homologous series of compounds containing up to four carbons of other functional groups:
HCHO, CH3CHO, CH3CH2CHO, CH3CH2CH2CHO
CH3COCH3, CH3COC2H5, CH3COCH2CH2CH3
HCOOH, CH3COOH, CH3CH2COOH,CH3CH2CH2COOH
Conclusion:
The common difference in the molecular formulae and molecular masses of any two successive (adjacent members) of a homologous series are CH2 unit and 14 u (mass units) respectively.
Activity 4.3 (Textbook Page 69)
Observation: Alcohol burns with a blue luminous flame and there is no sooty deposit on the metal plate. Thus, alcohol is a saturated compound.
In contrast, camphor and naphthalene burn with a yellow flame with lots of black smoke. Therefore, camphor and naphthalene are unsaturated compounds.
Conclusion: Saturated compounds burn with a luminous blue flame while unsaturated compounds burn with a yellow sooty flame.
Activity 4.4 (Textbook Page 69)
Observation and conclusion: When the hole of the Bunsen burner is closed, enough supply of air is not available and hence the gas bums incompletely to produce a yellow sooty flame.
However, when the hole is open, complete combustion of gas occurs and a blue flame is produced.
Activity 4.5 (Textbook Page 70)
Observation and conclusion: When alkaline KMnO4 is added to warm alcohol drop by drop, the colour of KMnO4 disappears because it oxidizes ethanol to ethanoic acid and is itself reduced to manganese dioxide (MnO2). On the other hand if KMnO4 solution added is more than what is required to oxidize ethanol to ethanoic acid, then some of the KMnO4 solution remains unused and hence its colour will not disappear.
Activity 4.6 (Textbook Page 72)
Observation and conclusion: When a small piece of sodium is added to absolute alcohol, a brisk effervescence occurs due to the evolution of H2 gas. Hydrogen is a combustible gas, therefore, when a burning splinter is brought near it, it burns with a pop sound.
Activity 4.7 (Textbook Page 73)
Observation: Dilute HCl and dilute CH3COOH turn blue litmus red, but this cannot decide that whether hydrochloric acid is a stronger acid than acetic acid.
But when we compare the pH of the aqueous solution of these two acids using universal indicator, we find that the pH of dilute acetic acid is around 4 (yellowish pink colour) while that of hydrochloric acid is around 2 (light pink colour). Therefore, acetic acid is a weaker acid than hydrochloric acid.
Conclusion: Since HCl is completely ionised while acetic acid is only partly ionised, therefore, hydrochloric acid is a stronger acid than acetic acid.
Activity 4.8 (Textbook Page 73)
Observation and conclusion: Absolute ethanol reacts with glacial acetic acid in presence of a few drops of conc. H2SO4 to form a sweet-smelling ester, i.e., ethyl ethanoate. Conc. H2SO4 removes the H2O formed during the reaction and shift the equilibrium in the forward direction.
Activity 4.9 (Textbook Page 74)
Observation and conclusion: When sodium carbonate is added to a dilute solution of ethanoic acid, a brisk effervescence is produced due to the evolution of carbon dioxide (CO2) gas.
When CO2 gas is passed through freshly prepared lime water, it turns lime water milky due to the formation of insoluble calcium carbonate.
Same observations are made when sodium bicarbonate is used instead of sodium carbonate.
Activity 4.10 (Textbook Page 74)
Observation and conclusion: When test tube B containing cooking oil, water and soap is allowed to stand after vigorous shaking only one layer was observed. This shows that oil dissolves in soap and thus helps in cleaning.
When test tube A containing water and cooking oil was allowed to stand after vigorous shaking, two separate layers – one of water and the other of oil was seen. This shows that oil does not dissolve in water and hence water alone cannot clean clothes.
Activity 4.11 (Textbook Page 76)
Observation and conclusion: The test tube that contains soft water, i.e., distilled water or rainwater, produces foam readily. On the other hand, the test tube which contain hard water produces a curdy white precipitate due to the formation of insoluble calcium or magnesium salts.
Activity 4.12 (Textbook Page 76)
Observation: Both the test tube do not have the same amount of foam/lather. The test tube in which soap solution was added produced a curdy white precipitate with little or no foam at all, while the tube containing detergent produced a large amount of foam with little or no white precipitate.
Conclusion: Detergent can be used for washing both in soft and hard water while soaps can be used only in soft water.